Technology
(paper)
Bringing cellular clarity to the cortical component of ALSwith a high-density multi-electrode array system
FEBS J. (2026). DOI: 10.1111/febs.70552
2026
Keywords:
There is selective vulnerability in diseases, requiring the understanding ofcell-type specific aspects of neurodegeneration with cellular resolution.Single-cell electrophysiology enables direct investigation of neuronal excit-ability, ion-channel dynamics, and synaptic transmission with high tempo-ral precision, which is crucial in understanding neuronal circuitries andhow they are affected in diseases. Microelectrode arrays (MEAs) haveemerged as powerful platforms enabling long-term, parallel, andnon-invasive extracellular measurements. The advent of complementary-metal-oxide-semiconductor (CMOS)-based and high-density microelectrodearrays (HD-MEAs) has expanded the spatial and temporal resolutionattainable both in vitro and ex vivo. Combined with acute slices, novelcell-culture approaches and three-dimensional (3D) brain organoids, thesetools now expedite translational research in disease modeling, neurotoxic-ity, and pharmacology. Here, we summarise the significance of single-cellelectrophysiology, the advantages of the MEA systems, and the latest bio-medical and technological advances in this area of research.

Organoids & Spheroids
(paper)
Human microglia in brain assembloids display region-specific diversity and respond to hyperexcitable neurons carrying SCN2A mutation
Sci. Adv. (2026). DOI: 10.1126/sciadv.ady2977
2026
Keywords:
Microglia critically shape neuronal circuit development and function, yet their region-specific properties and roles in distinct circuits of the human brain remain poorly understood. In this study, we generated region-specific brain organoids (cortical, striatal, and midbrain), each integrated with human microglia, to fill this critical gap. Single-cell RNA sequencing uncovered six distinct microglial subtypes exhibiting unique regional signatures, including a subtype highly enriched for the GABAB receptor gene within striatal organoids. To investigate the contributions of microglia to neural circuitry, we created microglia-incorporated midbrain-striatal assembloids, modeling a core circuit node for many neuropsychiatric disorders, including autism. Using chemogenetics to activate this midbrain-striatal circuit, we observed increased calcium signaling in microglia involving GABAB receptors. Leveraging this model, we examined microglial responses within neural circuits harboring an SCN2A nonsense (C959X) mutation associated with profound autism. Microglia displayed heightened calcium responses to SCN2A mutation–mediated neuronal hyperactivity and engaged in excessive synaptic pruning. These pathological effects were reversed not only by pharmacological inhibition of microglial GABAB receptors but also by knockout of the GABBR1 gene in microglia. Collectively, our findings establish an advanced platform that can be used to dissect human neuroimmune interactions in subcortical regions and to evaluate previously undiscovered therapies, highlighting the important role of microglia in shaping critical circuitry related to neuropsychiatric disorders.

Organoids & Spheroids
(paper)
Human neuromuscular organoids mimic cancer-induced muscle cachexia
Cell Rep. Methods (2026). DOI: 10.1016/j.crmeth.2026.101331
2026
Keywords:
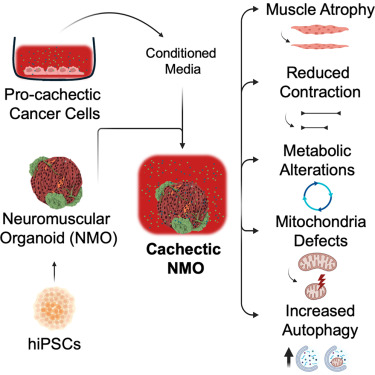
Motivation
The study of mechanisms behind cancer-induced muscle cachexia is a significant challenge. Currently available in vivo and in vitro models are limited and do not allow the study of cachexia in a context that is relevant to patients. We therefore sought to develop a multicellular, three-dimensional (3D) in vitro model derived from human cells that recapitulates the phenotypes observed in a cachectic muscle, including impairment of muscle contraction and cellular and molecular alterations.
Highlights
· hiPSC-derived NMOs mature andrespond to genetically induced muscle atrophy
· NMOs mimic cachectic musclemorphology and function under pro-cachectic factors
· Cachectic NMOs show cancercachexia metabolic, mitochondrial, and molecular features
· NMOs offer a platform to studymechanisms and test potential cachexia therapies
Summary
Cancer cachexia, a devastating metabolic wasting syndrome affecting up to 80% of solid cancer patients, remains incurable despite advances in tumor biology understanding. This study introduces neuromuscular organoids (NMOs) derived from human-induced pluripotent stem cells (hiPSCs) as a platform to investigate cancer-driven muscle cachexia. We found that NMOs respond well to atrophic stimuli and replicate the key features of cancer cachexia when treated with conditioned media derived from cachexia-inducing cancer cells. Specifically, cachectic NMOs showed muscle mass loss, impairment of muscle contraction, alteration of intracellular calcium homeostasis, appearance of mitochondrial dysfunction with a metabolic shift, and enhancement of autophagy. Based on these results, we propose NMOs derived from hiPSCs as an in vitro tool for investigating human muscle cachexia, with potential future avenues of patient-specific modeling and therapeutic screening.
.png)
Neuronal Cultures
(paper)
Microscale dysfunction and mesoscale compensation in degenerating neuronal networks
Netw. Neurosci. (2026). DOI: 10.1162/NETN.a.552
2026
Keywords:
Progressive neurodegenerative diseases involve neuronal dysfunction across cellular, circuit, and whole-brain levels. Despite differences in anatomical origins, vulnerable neuronal subtypes, and specific misfolded proteins, these diseases share key features. In presymptomatic phases, neural networks engage compensatory processes to maintain function, including increased centralisation and reliance on a rich-club of hub nodes. While such mechanisms have supporting evidence in some disorders, they remain less established in ALS, limiting understanding of potential shared pre-symptomatic responses. To address this, we investigated structural and functional properties of ALS patient-derived motor neuron networks compared to healthy controls using longitudinal multielectrode array recordings and graph theory-based analysis. We observed microscale dysfunction marked by TDP-43 proteinopathy, hyperactivity, and reduced spike amplitude. Structurally, ALS networks exhibited neurite hypertrophy, suggesting attempts to form new connections. Mesoscale analyses revealed functional reconfigurations, including increased rich-club connectivity and network assortativity, indicating compensatory centralisation. Our findings provide novel evidence that ALS network features can be recapitulated in in vitro models, and that these networks progressively become more centralised to preserve computational capacity, imposing growing demands on hub nodes and predisposing them to further damage. These results support models proposing common network reconfiguration mechanisms across neurodegenerative diseases.

Organoids & Spheroids
(paper)
Multi-omic analysis of guided and unguided forebrain organoids reveals differences in cellular composition and metabolic profiles
Cell Rep. Methods (2026). DOI: 10.1016/j.crmeth.2025.101295
2026
Keywords:
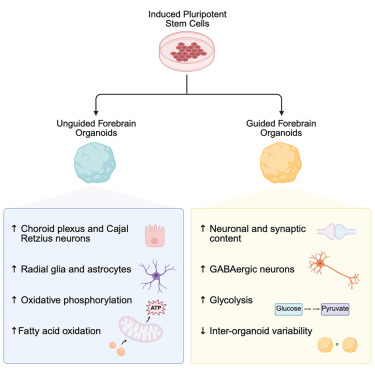
Highlights
• We present a broad multi-omic analysis of guided and unguided forebrain organoids
• We demonstrate significant differences in neuronal and glial cell type compositions
• Guided and unguided forebrain organoids have different metabolic profiles
• Modeling of disease-relevant phenotypes may be influenced by choice of organoid method
Summary
Neural organoids are invaluable model systems for studying neurodevelopment, generated by either guided or unguided approaches. Despite the importance for the field, the resulting differences between these models are unclear. To obtain an unbiased comparison, we performed a multi-omic analysis of forebrain organoids generated in parallel with two widely applied guided and unguided protocols. The guided forebrain organoids contained a larger proportion of neurons, including GABAergic interneurons, whereas the unguided organoids contained significantly more choroid plexus, radial glia, and astrocytes at later stages. Substantial differences in metabolic profiles were identified, pointing to increased levels of oxidative phosphorylation and fatty acid β-oxidation in the unguided forebrain organoids and a higher reliance on glycolysis in the guided forebrain organoids. Overall, our study comprises a thorough description of the multi-omic differences between these guided and unguided forebrain organoids and provides an important resource for the neural organoid field studying neurodevelopment and disease.
Neuronal Cultures
(paper)
Protocol for tailored in vitro neuronal networks on high-density microelectrode arrays with polydimethylsiloxane microstructures
STAR Protoc. (2026). DOI: 10.1016/j.xpro.2026.104349
2026
Keywords:

Complementary metal-oxide-semiconductor (CMOS)-based high-density microelectrode arrays (HD-MEAs) enable neuronal recordings with high spatiotemporal resolution. However, integrating polydimethylsiloxane (PDMS) microstructures onto HD-MEA surfaces to control network architecture is currently challenging and platform specific. Here, we present a protocol for PDMS fabrication, HD-MEA chip preparation, PDMS-HD-MEA microstructure alignment, and cell culture, including alternatives. Our results show reproducible formation of modular networks with characteristic activity patterns across different systems. This protocol supports engineering of defined neuronal architectures while maintaining compatibility with various HD-MEA systems.

Organoids & Spheroids
(paper)
Intermittent Propofol Exposure Induces Neurodevelopmental Alterations in Human Brain Organoids
Cell. Mol. Neurobiol. (2026). DOI: 10.1007/s10571-026-01673-2
2026
Keywords:
The administration of anaesthesia during pregnancy may have implications for fetal brain development. This study used H1 embryonic stem cell-derived human brain organoids (HBOs) to investigate effects of intermittent propofol exposure (IPE). HBOs were subjected to early IPE from 47 to 50 days in vitro (div), or late IPE from 77 to 80 div, using a clinically supra-anaesthetic concentration of 50 µM propofol. This was followed by cultivation without propofol for an additional 10 div, and HBOs were subsequently analysed at 60 or 90 div. Determination of HBO growth and lactate release did not provide evidence of neurotoxicity. Multi-electrode array recordings indicated an increased neuronal activity at 60 div following early IPE, an effect not observed at 90 div following late IPE. RNA-sequencing revealed that IPE up-regulated genes associated with neurodevelopment and synapse functions at 60 div, which overlapped with naturally up-regulated genes during HBO development from 60 to 90 div. These findings indicate that early IPE accelerates brain maturation in HBOs, suggesting possible deviations from the normal developmental trajectory in the fetal brain.

Acute Brain Slices
(paper)
Organotypic human brain slice cultures as a translational testing platform for novel neuromodulation devices
J. Neural Eng. (2026). DOI: 10.1088/1741-2552/ae34e9
2026
Keywords:
To establish organotypic human brain slice cultures (hBSCs) as a translational screening platform for evaluating novel neuromodulation devices and to demonstrate the feasibility of the model using magnetoelectric nanoparticles (MENPs) as a representative neurostimulation modality.
Approach: Viable hBSCs were prepared from resected cortical tissue of epilepsy surgery patients and a GCaMP-based calcium imaging, multi-electrode array (MEA) recordings, and immunohistochemical staining for c-Fos was conducted. The MENPs were injected into the hBSCs and stimulated with an alternating magnetic field to assess their neuromodulatory effects.
Main Results: GCaMP transduction enables the real-time visualization of MENP-induced neuronal activity. Electrophysiological signals, including spiking and local field potentials, were observed in fresh, but not cultured, slices. c-Fos immunostaining revealed a significant increase in c-Fos expression in stimulated MENP-injected cultures compared to sham-treated controls. This protocol yielded reproducible tissue viability and consistent results across patient-derived samples.
Significance: This technical note demonstrates that hBSCs represent a reproducible and ethically preferable translational model suitable for screening applications in neurotechnology research. The platform enables early-stage functional evaluation of neuromodulatory devices, particularly those with a higher risk of failure in vivo or curiosity-driven early-phase concepts in a setting superior to traditional in vitro approaches. This platform may help reduce reliance on animal models in neurotechnology development.

Organoids & Spheroids
(paper)
Targeted antisense oligonucleotide treatment rescues developmental alterations in spinal muscular atrophy organoids
Nat. Commun. (2025). DOI: 10.1038/s41467-025-67725-1
2025
Keywords:
Spinal muscular atrophy (SMA) is a severe neurological disease caused by mutations in the SMN1 gene, characterized by early onset and degeneration of lower motor neurons. Understanding early neurodevelopmental defects in SMA is crucial for optimizing therapeutic interventions. Using spinal cord and cerebral organoids generated from multiple SMA type 1 male donors, we revealed widespread disease mechanisms beyond motor neuron degeneration. Single-cell transcriptomics uncovered pervasive alterations across neural populations, from progenitors to neurons, demonstrating SMN-dependent dysregulation of neuronal differentiation programs. Multi-electrode array (MEA) analysis identified consistent hyperexcitability in both spinal and brain organoids, establishing altered electrical properties as a central nervous system-wide feature of pathogenesis. Early administration of an optimized antisense oligonucleotide (ASO) that increased SMN levels rescued morphological and functional deficits in spinal cord organoids across different genetic backgrounds. Importantly, this early intervention precisely corrected aberrant splicing in here identified SMN1 targets enriched at critical nodes of neuronal differentiation. Our findings demonstrate that early developmental defects are core features of SMA pathogenesis that can be prevented by timely therapeutic intervention, providing insights for optimizing treatment strategies.

Acute Brain Slices
(conf. proc.)
Network-Level Characterization of Hippocampal Disruptions in Alzheimer’s Disease Using Large-Scale Electrophysiology
IEEE EMBC (2025). DOI: 10.1109/EMBC58623.2025.11253272.
2025
Keywords:
Alzheimer’s disease (AD), a progressive neurodegenerative disorder, is projected to affect over 130 million people globally by 2050. While extensive efforts have focused on targeting molecular hallmarks such as amyloid-beta (Aβ) plaques and tau pathology, network-level dysfunction remains a critical but underexplored component of AD progression. Disruptions in hippocampal-cortical (HC) circuit activity emerge early in AD, compromising memory processing and cognitive functions. Characterizing these disruptions requires high-resolution platforms capable of capturing network-wide spatiotemporal dynamics. To address this, we implemented a high-density microelectrode array (HD-MEA) biosensor to assess large-scale electrophysiological activity in ex vivo hippocampal slices from well-established APPNL and APPNL-G-F mouse models. Our approach quantifies hippocampal oscillatory disturbances and examines their modulation by saffron, a natural compound with reported neuroprotective properties. Results indicate that hippocampal network activity is progressively impaired in APPNL-G-F mice, particularly in sharp-wave ripple (SWR) and multi-unit activity (MUA) patterns. The HD-MEA platform provides a scalable tool for investigating AD-associated network dysfunctions and exploring potential modulatory interventions.
Neuronal Cultures
(paper)
Protocol for the enhanced analysis of electrophysiological data from high-density multi-electrode arrays with nicespike and spikeNburst
STAR Protoc. (2025). DOI: /10.1016/j.xpro.2025.104195
2025
Keywords:

High-density multi-electrode arrays enable the recording of in vitro neuronal activity with exceptional spatial and temporal resolution. Here, we describe a protocol for analyzing these extensive datasets by using two complementary tools. The nicespike tool implements a full electrophysiological data analysis pipeline featuring graphics processing unit–accelerated spike sorting via template matching with Kilosort, enabling accurate identification of neuronal units across multiple electrodes. The spikeNburst tool incorporates advanced techniques for spike train filtering, burst and network burst detection, and synchronization analysis.

Acute Brain Slices
(paper)
High-density multielectrode array (MEA) recordings in a rodent glioma model
J. Neurosci. Methods (2025). DO: /10.1016/j.jneumeth.2025.110623
2025
Keywords:
• Network bursts were induced in acute brain slices from glioma-bearing rats.
• 64 × 64 multielectrode arrays allowed for detailed recording of network bursts.
• Center-of-activity trajectories and activity propagation analysis were described.
• Network bursts differed between glioma-bearing slices and controls.
• Spike Time Tiling Coefficient-based functional connectivity graphs were constructed.

Organoids & Spheroids
(paper)
Generation of human nucleus basalis organoids with functional nbM-cortical cholinergic projections in transplanted assembloids
Cell Stem Cell (2025). DOI: 10.1016/j.stem.2025.10.004
2025
Keywords:
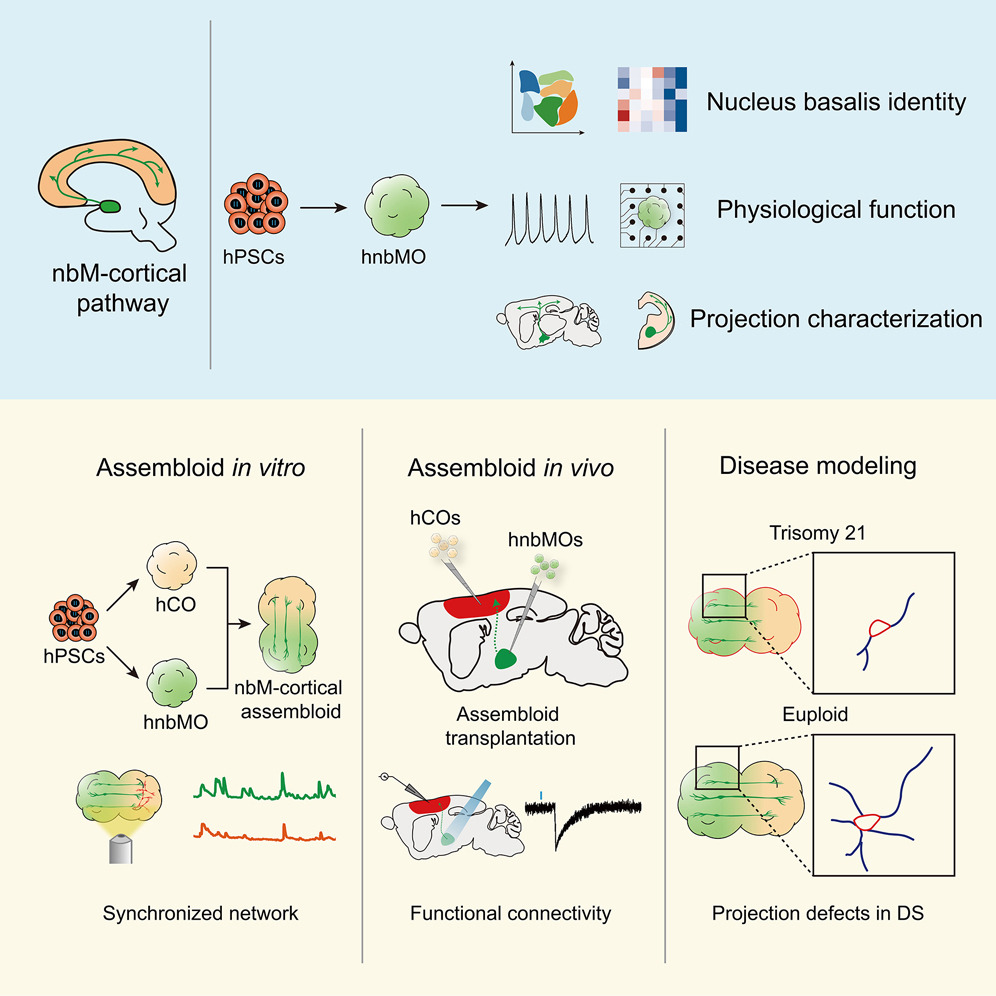
Highlights
• Human nbM organoids generated from hPSCs recapitulate nucleus-specific identity
• hnbMOs establish long projection and functional connectivity with hCOs in vitro
• Assembloid transplantation formed the functional nbM-cortical cholinergic pathway
• nbM-cortical assembloids model projection deficits in Down syndrome
Summary
The nucleus basalis of Meynert (nbM), the major cholinergic output of the basal forebrain, regulates cortical modulation, learning, and memory. Dysfunction of the nbM-cortical cholinergic pathway is implicated in neurodegenerative and neurodevelopmental disorders, including Alzheimer’s disease (AD) and Down syndrome (DS). Here, we generated human nbM organoids (hnbMOs) from human pluripotent stem cells (hPSCs) containing functional cholinergic projection neurons. Then we reconstructed long-distance cholinergic projections from nbM to the cortex by co-culturing hnbMOs with human fetal brains and transplanting hnbMOs into immunodeficient mice. We further established nbM-cortical assembloids by fusing hnbMOs with human cortical organoids (hCOs). We also established a human-specific cholinergic projection system in transplanted assembloids. Using viral tracing and functional assays, we validated that cholinergic neurons send projections into hCOs and form synaptic connections. Moreover, we captured projection deficits in DS-derived assembloids, demonstrating the utility of this model for studying nbM-related neural circuits and neurological disorders.

Organoids & Spheroids
(paper)
High Resolution Detection of Stationary and Evolving 2D-Current Source Density within Neuronal Microcircuits
J Neurophysiol. (2025). DOI: 10.1152/jn.00098.2025
2025
Keywords:
The use of a high-density microelectrode arrays (>4000 microelectrodes) allows the recordings of evoked and on-going field activity of the whole hippocampal formation and other tissues with high spatiotemporal resolution. From these recordings it is possible to obtain the current source density (CSD) which separates the current generators into sinks and sources; this differentiation provides the means to distinguish correlated, disjoint loci of activity and track them separately at near to cell resolution. By obtaining the vectorial average of an area regarded as sink or source, we obtain a putative center of action, or center of mass that can be traced in time. Thus, successive centers would reveal the immediate nearby correlated units that "inherit" or otherwise are affected by this activity revealing a putative route of transfer of information that can be quantitated with high spatiotemporal definition. For structured tissues, this methodology provides a means to infer effective information transmission with clear parameters that can be further analyzed in several ways. Importantly, the CSD over time reveals patterns of activity obscured by the representation of activity in the voltage domain, which can aid to uncover synaptic interactions in restricted microcircuits.

Neuronal Cultures
(paper)
From Neural Activity to Computation: Biological Reservoirs for Pattern Recognition in Digit Classification
ICCVW (2025). DOI: 10.48550/arXiv.2510.05637
2025
Keywords:
In this paper, we present a biologically grounded approach to reservoir computing (RC), in which a network of cultured biological neurons serves as the reservoir substrate. This system, referred to as biological reservoir computing (BRC), replaces artificial recurrent units with the spontaneous and evoked activity of living neurons. A multi-electrode array (MEA) enables simultaneous stimulation and readout across multiple sites: inputs are delivered through a subset of electrodes, while the remaining ones capture the resulting neural responses, mapping input patterns into a high-dimensional biological feature space. We evaluate the system through a case study on digit classification using a custom dataset. Input images are encoded and delivered to the biological reservoir via electrical stimulation, and the corresponding neural activity is used to train a simple linear classifier. To contextualize the performance of the biological system, we also include a comparison with a standard artificial reservoir trained on the same task. The results indicate that the biological reservoir can effectively support classification, highlighting its potential as a viable and interpretable computational substrate. We believe this work contributes to the broader effort of integrating biological principles into machine learning and aligns with the goals of human-inspired vision by exploring how living neural systems can inform the design of efficient and biologically plausible models.

Neuronal Cultures
(paper)
A proper excitatory/inhibitory ratio is required to develop synchronized network activity in mouse cortical cultures
Stem Cell Rep. (2025). DOI: 10.1016/j.stemcr.2025.102646
2025
Keywords:
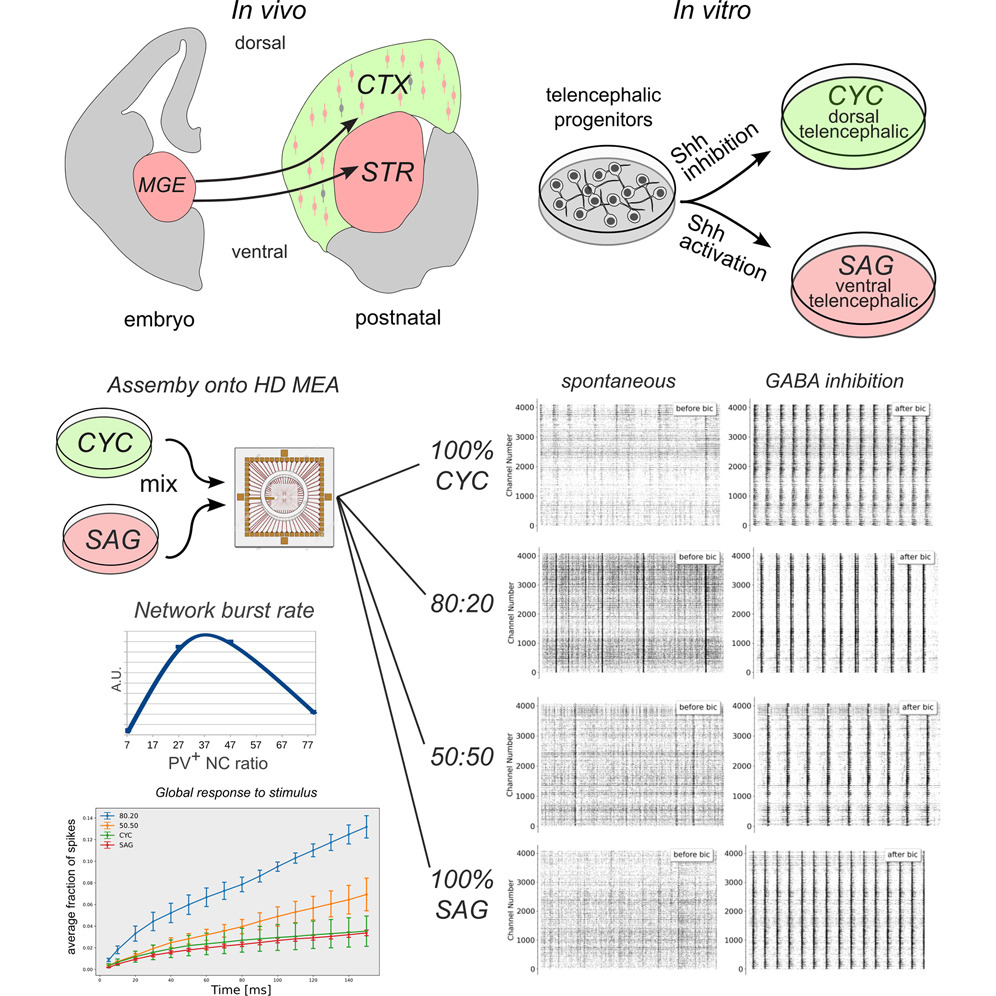
Highlights
• Cortical activity development relies on an optimal excitatory/inhibitory neuron balance
• In vitro modeled cortical networks reveal varied spontaneous activity patterns
• Inhibitory neurons critically influence bursting activity development
• Intermediate levels of E/I balance are required for network synchronized activity
Summary
Excitatory/inhibitory (E/I) balance is thought to play a key role in cortical activity development. We modeled an in vitro cortical network deployed of the inhibitory neurons normally migrating from the ventral telencephalon and implemented ventral telencephalic (VT) cultures and co-cultures with mixed proportions of dorsal telencephalic (DT) and VT neurons, containing distinct proportions of inhibitory neurons. Interestingly, these pure and mixed cultures developed different patterns of spontaneous activity and functional connectivity. Our findings highlighted a critical role for the inhibitory component in developing correlated network activity. Unexpectedly, networks with 7% of parvalbumin (PV)+ neurons were not able to generate appreciable network burst activity due to the development of a strong network inhibition, despite their lowest E/I ratio. Our observations support the notion that an optimal ratio of PV+ neurons during cortical development is essential for the establishment of local inhibitory networks capable of generating and spreading correlated activity.

Acute Brain Slices
(paper)
Enhanced electrophysiological recordings in acute brain slices, spheroids, and organoids using 3D high-density multielectrode arrays
PLoS One (2025). DOI: 10.1371/journal.pone.0328903
2025
Keywords:
Recent advances in three-dimensional (3D) biological brain models in vitro and ex vivo are creating new opportunities to understand the complexity of neural networks but pose the technological challenge of obtaining high-throughput recordings of electrical activity from multiple sites in 3D at high spatiotemporal resolution. This cannot be achieved using planar multi-electrode arrays (MEAs), which contact just one side of the neural structure. Moreover, the specimen adhesion to planar MEAs limits fluid perfusion along with tissue viability and drug application. Here, the efficiency of the tissue-sensor interface provided by advanced 3D high-density (HD)-MEA technology was evaluated in acute brain slices, spheroids, and organoids obtained from different brain regions. The 3D HD-MEA microneedles reached the inner layers of samples without damaging network integrity and the microchannel network between microneedles improved tissue vitality and chemical compound diffusion. In acute cortico-hippocampal and cerebellar slices, signal recording and stimulation efficiency proved higher with the 3D HD-MEA than with a planar MEA improving the characterization of network activity and functional connectivity. The 3D HD-MEA also resolved the challenge of recording from brain spheroids as well as cortical and spinal organoids. Our results show that 3D HD-MEA technology represents a valuable tool to address the complex spatiotemporal organization of activity in brain microcircuits, making it possible to investigate 3D biological models.

Acute Retina
(paper)
Saltatory axonal conduction in the avian retina
J. Physiol. (2025). DOI: /10.1101/2022.07.04.498722
2025
Keywords:
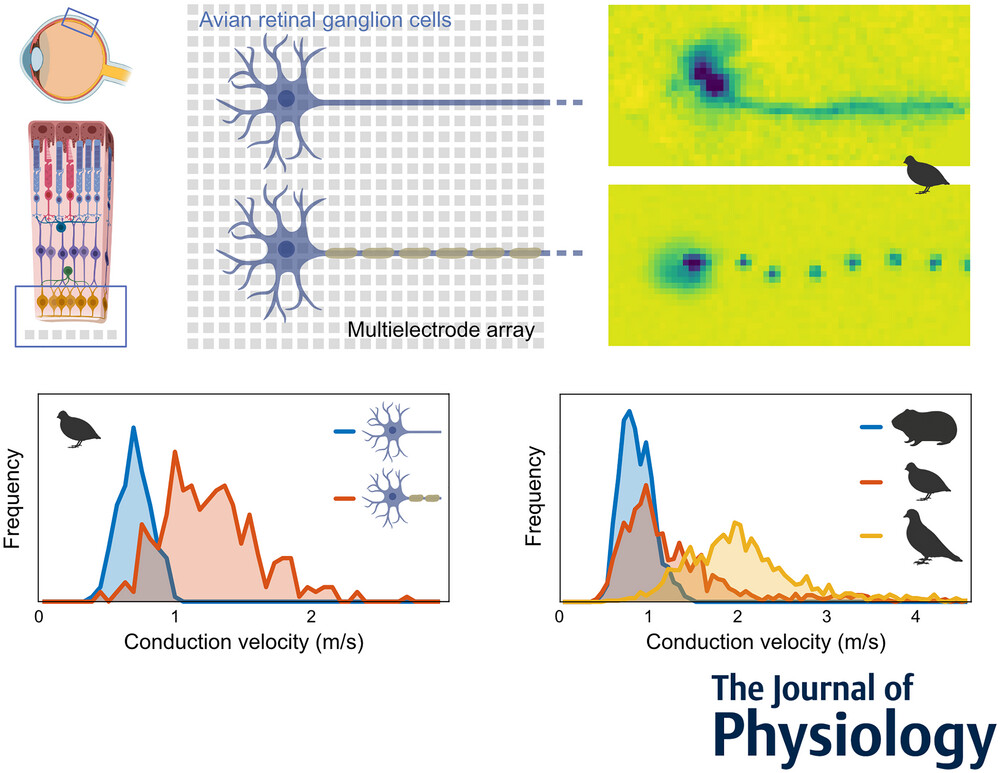
In contrast to most parts of the vertebrate nervous system, ganglion cell axons in the retina typically lack myelination. In the majority of species, ganglion cell axons only become myelinated after leaving the retina to form the optic nerve. The avian retina, however, presents a remarkable exception in that ganglion cell axons are partly myelinated in the retinal nerve fibre layer. It was hypothesized that the optically detrimental properties of retinal myelination are evolutionarily offset by advantages in spike conduction velocity. Using high-resolution multielectrode array recordings, we analysed the spike conduction in the retina of various avian species in comparison to mammalian species. Indeed, mammals showed lower conduction velocities than avian species. Myelinated axons typically achieved higher conduction velocities than unmyelinated axons. Notably, some myelinated axons exhibited conduction velocities lower than those of unmyelinated axons. Anatomical analyses revealed that myelination in the nerve fibre layer was accompanied by the formation of nodes of Ranvier. The internode length was positively correlated with the axon diameter. In physiological recordings, the spatial extent of simultaneously active nodes was positively correlated with the conduction velocity. Conversely, the internode length and the activation kinetics of a node were weak predictors of conduction velocity. Overall, this study illuminates the unique features of the avian retina and offers insights into the functional requirements and evolutionary pressures of myelination affecting conduction velocity in the nervous system.
Key points
- Intraretinal saltatory axonal spike conduction was studied across multiple avian species using high-resolution multielectrode arrays.
- The highest conduction velocities were observed exclusively in saltatory axons, while the lowest were found in non-saltatory axons. However, slow myelinated axons exist, exhibiting a surprisingly large overlap in conduction velocities with unmyelinated axons.
- The spatial extent of a spike showed a strong positive correlation with conduction velocity.
- The internodal length exhibited a positive correlation with axon diameter, and the variability in internodal length was smaller within individual axons than across axons.
- Intraretinal axonal conduction velocities across species appear to align with their ecological niche. The maximal intraretinal spike conduction velocity observed in birds was up to four times faster than in rodents.

Acute Brain Slices
(paper)
Unsupervised pipeline for the identification of cortical excitatory and inhibitory neurons in high-density multielectrode arrays with ground-truth validation
eLife (2025). DOI: 10.7554/eLife.106557.3
2025
Keywords:
Large-scale extracellular recording techniques have advanced the study of neuronal circuits but lack methods to reliably identify cell types while scaling to thousands of neurons. We introduce spikeMAP, a pipeline for analyzing large-scale in vitro cortical recordings that combines spike sorting with cell-type identification using viral and optogenetic validation. SpikeMAP integrates data analysis with optogenetic, viral, and pharmacological protocols to dynamically probe distinct cell types while recording from large populations. The pipeline fits spike waveforms using spline interpolation to measure half-amplitude and peak-to-peak durations, applies principal component analysis and k-means clustering to isolate single-neuron signals, and uses linear discriminant analysis to optimize cluster separability. Channel source locations are determined through spatiotemporal spike waveform characteristics. Applied to mouse prefrontal cortex slices recorded on a 4096-channel array, spikeMAP effectively distinguishes regular-spiking excitatory neurons from fast-spiking inhibitory interneurons via action potential waveform, Fano factor, and spatial cross-correlations. This validated toolbox enables comprehensive characterization of neuronal activity across cell types in high-density recordings, offering a scalable approach to study microcircuit interactions in the brain.
Acute Brain Slices
(paper)
Transplanted human striatal progenitors exhibit functional integration and modulate host circuitry in a Huntington’s disease animal model
Pharmacological Research (2025). DOI: 10.1016/j.phrs.2025.107905.
2025
Keywords:
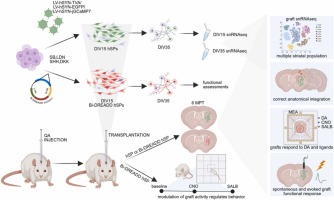
Huntington’s disease (HD) is a fatal neurodegenerative disorder caused by a CAG repeat expansion in the HTT gene. This leads to progressive loss of striatal neurons and motor-cognitive decline. While current gene-targeting approaches aiming at reducing somatic instability show promise – especially in case of early treatment – they cannot restore the already compromised neuronal circuitry at advanced disease stages. Thus, cell replacement therapy offers a regenerative strategy to rebuild damaged striatal circuits. Here, we report that human striatal progenitors (hSPs) derived from embryonic stem cells via a morphogen-guided protocol survive long-term when transplanted into a rodent model of HD and recapitulate key aspects of ventral telencephalic development. By employing single-nucleus RNAseq of the grafted cells, we resolved their transcriptional profile with unprecedented resolution. This has identified transcriptional signals of D1- and D2-type medium spiny neurons (MSN), Medial Ganglionic Eminence (MGE) and Caudal Ganglionic Eminence (CGE) -derived interneurons, and regionally specified astrocytes. Moreover, we demonstrate that grafted cells undergo further maturation 6 months post-transplantation, acquiring the expected regionally defined transcriptional identity. Immunohistochemistry confirmed stable graft composition over time and supported a neurogenic-to-gliogenic switch post-transplantation. Multiple complementary techniques including virus-based tracing and electrophysiology assays demonstrated anatomical and functional integration of the grafts. Notably, chemogenetic modulation of graft activity regulated striatal-dependent behaviors, further supporting effective graft integration into host basal ganglia circuits. Altogether, these results provide preclinical evidence that hSP-grafts can reconstruct striatal circuits and modulate functionally relevant behaviors. The ability to generate a scalable, molecularly defined progenitor population capable of in vivo functional integration supports the potential of hSPs for clinical application in HD and related basal ganglia disorders.

Acute Brain Slices
(paper)
Dynamic mapping of network-level LTP in the hippocampus via high-resolution bioelectrical sensing
APL Bioeng. (2025). DOI: /10.1063/5.0258985
2025
Keywords:
Understanding the complexity of neural network dynamics demands advanced biosensing technologies capable of capturing large-scale interactions with high spatial and temporal precision. Traditional approaches, such as patch-clamp and field recordings, are inherently limited in resolving network-wide synaptic connections, particularly long-term potentiation (LTP), due to their localized scope and indirect access to hippocampal subfields. To address these challenges, we introduce EvoNES, a CMOS-based high-definition 4096 microelectrode array platform that leverages bidirectional stimulus-responsive biosensing functionality. By coupling precise external electrode stimulation targeting the Schaffer collateral and medial perforant pathways with simultaneous on-chip bioelectrical recordings, EvoNES enables the first real-time quantification of evoked responses and LTP dynamics across the entire hippocampal circuit. This system bridges critical gaps in traditional techniques, providing a mesoscopic-scale view of cell assemblies interplay and delivering unprecedented insights into the distributed mechanisms underlying memory encoding and learning processes. Advanced computational analyses generate variation maps revealing distinct voltage fluctuation patterns and differential sensitivity across hippocampal subregions during synaptic potentiation. Our findings identify four distinct waveform classes within the CA1–CA3 network and three unique evoked firing patterns in the dentate gyrus (DG). Post-tetanic responses show faster induction, expanded activated zones, and the activation of previously silent cell assemblies, indicating significant network restructuring. Applied in aged mice, EvoNES demonstrates age-dependent changes in network LTP, both quantitatively and qualitatively. This high-resolution biosensing platform in a live neural context provides unprecedented insights into hippocampal memory formation and offers a powerful tool for investigating neural plasticity and network interactions in both health and disease states.

Neuronal Cultures
(paper)
The lncRNA Gas5 is an activity-responsive scaffold that mediates cAMP-dependent synaptic plasticity
Sci. Signal. (2025). DOI: 10.1126/scisignal.adn2044
2025
Keywords:
Changes in the transcriptome are critical in shaping the structural plasticity of neurons, which underpins learning and long-term memory storage. Here, we explored the effect of two opposing, plasticity-associated pathways—cAMP second-messenger signaling and metabotropic glutamate receptor (mGluR1 and mGluR5) signaling—on the transcriptome in hippocampal neurons and how these pathways operate in distinct and coordinated manners to induce structural changes. Integration of transcriptome data and molecular pathway analysis identified central “hub” genes that were rapidly induced by cAMP and/or mGluR1/5 in hippocampal neurons. These included the long noncoding RNA (lncRNA) Gas5, whose expression was induced specifically by cAMP and which was targeted to dendrites by the kinesin motor protein KIF1A. In the dendrites, Gas5 interacted with various proteins and coding and noncoding RNAs associated with synaptic function and plasticity, and these interactions were altered by cAMP signaling. Gas5 interacted with the microRNA miR-26a-5p and sequestered it from several of its mRNA targets associated with neuronal function and whose translation was induced by cAMP. Gas5 was critical for excitatory synaptic transmission induced by cAMP but not those induced by mGluR1/5. Furthermore, Gas5 deficiency impaired dendritic branching and synapse morphology, and Gas5 abundance was decreased in the hippocampus of a mouse model of Alzheimer’s disease. Together, these findings provide insight into the transcriptional networks involved in synaptic plasticity and a lncRNA interactome that mediates dendritically localized regulation of excitatory synaptic transmission and neuronal architecture.

Neuronal Cultures
(paper)
CACNA1A loss-of-function affects neurogenesis in human iPSC-derived neural models
Cell. Mol. Life Sci. (2025). DOI: 10.1007/s00018-025-05740-7
2025
Keywords:
CACNA1A encodes the pore-forming α1A subunit of the CaV2.1 calcium channel, whose altered function is associated with various neurological disorders, including forms of ataxia, epilepsy, and migraine. In this study, we generated isogenic iPSC-derived neural cultures carrying CACNA1A loss-of-function mutations differently affecting CaV2.1 splice isoforms. Morphological, molecular, and functional analyses revealed an essential role of CACNA1A in neurodevelopmental processes. We found that different CACNA1A loss-of-function mutations produce distinct neurodevelopmental deficits. The F1491S mutation, which is located in a constitutive domain of the channel and therefore causes a complete loss-of-function, impaired neural induction at very early stages, as demonstrated by changes in single-cell transcriptomic signatures of neural progenitors, and by defective polarization of neurons. By contrast, cells carrying the Y1854X mutation, which selectively impacts the synaptically-expressed CaV2.1[EFa] isoform, behaved normally in terms of neural induction but showed altered neuronal network composition and lack of synchronized activity. Our findings reveal previously unrecognized roles of CACNA1A in the mechanisms underlying neural induction and neural network dynamics and highlight the differential contribution of the divergent variants CaV2.1[EFa] and CaV2.1[EFb] in the development of human neuronal cells.

Acute Brain Slices
(paper)
Neurodevelopmental origins of structural and psychomotor defects in CXCR4-linked primary immunodeficiency
Neuron (2025). DOI: 10.1016/j.neuron.2025.05.016
2025
Keywords:
Inborn errors of immunity (IEI), as congenital chronic disorders, are often associated with neurobehavioral symptoms, traditionally considered secondary to patient burden. Their origin, however, has yet to be addressed. Here, we found that IEI-associated genes are expressed in neural lineages during human brain development, and in the absence of immunological challenges, IEI mutations directly impair neurodevelopmental trajectories, leading to psychomotor defects. Warts hypogammaglobulinemia immunodeficiency myelokathexis (WHIM) mice—bearing a mutation causing Cxcr4 hyperactivation—show developmental foliation defects of the cerebellum correlating with sensorimotor and affective dysfunctions, which recapitulate the alterations described in patients. WHIM cerebella single-cell profiling revealed major transcriptional deregulation in granule cell progenitors, whose aberrant proliferation and migration induce foliation and circuit defects. AMD3100 intracerebroventricular injection rescues both morphological and behavioral defects, demonstrating their brain-specific and Cxcr4-dependent origin. Collectively, our findings highlight the relevance of neurodevelopmental implications underlying psychomotor IEI manifestations, broadening our understanding of these conditions beyond immune dysfunctions.

Acute Retina
(paper)
Filter-based models of suppression in retinal ganglion cells: Comparison and generalization across species and stimuli
PLoS Comput Biol (2025). DOI: /10.1371/journal.pcbi.1013031
2025
Keywords:
The dichotomy of excitation and suppression is one of the canonical mechanisms explaining the complexity of neural activity. Computational models of the interplay of excitation and suppression in single neurons aim at investigating how this interaction affects a neuron’s spiking responses and shapes the encoding of sensory stimuli. Here, we compare the performance of three filter-based stimulus-encoding models for predicting retinal ganglion cell responses recorded from axolotl, mouse, and marmoset retina to different types of temporally varying visual stimuli. Suppression in these models is implemented via subtractive or divisive interactions of stimulus filters or by a response-driven feedback module. For the majority of ganglion cells, the subtractive and divisive models perform similarly and outperform the feedback model as well as a linear-nonlinear (LN) model with no suppression. Comparison between the subtractive and the divisive model depends on cell type, species, and stimulus components, with the divisive model generalizing best across temporal stimulus frequencies and visual contrast and the subtractive model capturing in particular responses for slow temporal stimulus dynamics and for slow axolotl cells. Overall, we conclude that the divisive and subtractive models are well suited for capturing interactions of excitation and suppression in ganglion cells and perform best for different temporal regimes of these interactions.